It can be installed in a shorter time, at a lower cost, and in a smaller space than manufacturing a clean room. Moreover, the cleanliness can be locally enhanced and can be up to Class 100. The clean room can be designed freely to suit the customer's work environment, from a simple, simplified type to temperature control that takes into account exhaust heat from the equipment, UV-cut specifications, air showers, pass boxes, and more. This system prevents product contamination, improves quality control, and enables a stable and efficient product supply.

Cleanroom
We provide tailor-made clean rooms with cleanliness levels from Class 1 to 100,000 to eliminate contaminants.
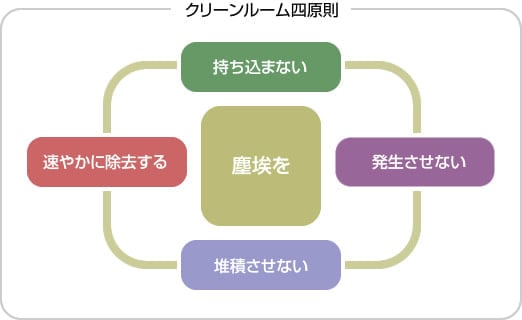
In clean rooms, cleanliness, temperature, humidity, room pressure, and the number of microorganisms (suspended and fallen bacteria) are controlled using air conditioning equipment. To maintain a clean environment in a clean room, the "Four Clean Room Principles" must be strictly adhered to.
Classification of Cleanrooms
Industrial clean rooms (industrial clean rooms) control physical dust floating in the air for the purpose of achieving high quality in the manufacture, assembly, and inspection of electronic devices, as typified by semiconductor manufacturing.
Biological clean rooms are used in pharmaceutical plants, hospitals, and other medical research facilities to remove bacteria, viruses, and other microorganisms that adhere to dust and float in the air.

Introduction and Selection
A clean environment is required.
Semiconductors and various electronic components are used in all kinds of electronic equipment, from AV equipment such as cell phones, PCs, and TVs to large supercomputers. In order to improve the quality and uniformity of precision, dust reduction is always required in the manufacturing environment for ultra-fine processing.
In the field of pharmaceuticals and cosmetics, there is a constant demand for improved quality, longer quality retention periods, and safety, and contamination by bacteria and microorganisms is the greatest concern in the regenerative medicine field, which is now attracting the most attention.
Clean rooms and clean booths are indispensable for these cutting-edge technologies and medical sites.

I just want to keep costs down!
We all want to start at a low cost. It is now easy to purchase simple booths that can be assembled by yourself through mail-order.
However, will a clean environment be maintained by using thin frames that seem to bend under load, or by using a booth without conducting the all-important cleanliness test after installation and measurement? Can it be maintained for a long time?
The best thing to do is to create a clean environment that matches the customer's requirements, rather than to match the environment to the size of a simple booth. We offer products that are different from standard products at low prices.

Planning - Construction
We can accommodate your environment.
You want a local booth to emphasize cleanliness.
You want to install a clean room that fits in with an existing room.
Your budget is limited, but you want specifications that meet your needs.
We are new to this, so we don't know where to start.
Please feel free to contact us at ・・・・ to discuss the installation space, purpose of use, budget, and operation schedule.
We will be happy to make a proposal to meet your needs in any field of work environment.


Cleanliness
The cleanliness of a clean room is called the cleanliness class, which in principle refers to the cleanliness at the completion of construction.
In the Japanese Industrial Standard (JIS), it is expressed in terms of a power index, which is the number of particles in 1 m3 of air expressed as a power of 10. In the long-used U.S. Federal Standard (USA Fed. Std. 209E), it is expressed as the number of particulates in 1 f3 (cubic foot ≈ 28.3 liters). The International Organization for Standardization (ISO) cleanliness class is almost entirely based on JIS B9920.
The following table shows a comparison of the relationship between the classifications of this standard and conventional standards.
| ISO cleanliness class |
Allowable particle concentration above specified particle size (pcs/m3) |
FED-STD-209D Cleanliness Class |
JISTarget particle size Range(μm) |
|||||
|---|---|---|---|---|---|---|---|---|
| 0.1μm | 0.2μm | 0.3μm | 0.5μm | 1μm | 5μm | |||
| 1 | 10 | 2 | - | - | - | 0.1~0.3 | ||
| 2 | 100 | 24 | 10 | 4 | - | - | 0.1~0.3 | |
| 3 | 1,000 | 237 | 102 | 35 | 8 | - | 1 | 0.1~0.5 |
| 4 | 10,000 | 2,370 | 1,020 | 352 | 83 | - | 10 | 0.1~0.5 |
| 5 | 100,000 | 23,700 | 10,200 | 3,520 | 832 | 29 | 100 | 0.1~5 |
| 6 | 1,000,000 | 237,000 | 102,000 | 35,200 | 8,320 | 293 | 1,000 | 0.3~5 |
| 7 | - | - | - | 352,000 | 83,200 | 2,930 | 10,000 | 0.3~5 |
| 8 | - | - | - | 3,520,000 | 832,000 | 29,300 | 100,000 | 0.3~5 |
| 9 | - | - | - | 35,200,000 | 8,320,000 | 293,000 | 1,000,000 | |